Opinion
Numbers behind different COVID-19 vaccines
By M. C. M. Iqbal
The vaccines against COVID-19, available today, are based on different strategies and come with different numbers to indicate their performance. Many of us wish to know if one vaccine is better than the other. Two concepts underlying the performance of the vaccines are efficacy and effectiveness. The Pfizer-BioNTech vaccine has an efficacy of 95 percent, the Moderna Vaccine is 94.5 percent and the Russian made Sputnik vaccine is over 90 percent. Does this mean some vaccines are better than the other? The short answer is no. All the approved vaccines are equally good. So, let us look at what these numbers mean.
These numbers refer to statistical calculations to interpret the results of vaccination trials conducted by the manufacturers of vaccines, following a prescribed format. The method of calculation was developed over 100 years ago by two statisticians, who published their results in the Proceedings of the Royal Society of Medicine in 1915. They, Major Greenwood (Major is his first name and not a military title) and Udny Yule, were tasked with interpreting the results of immunization of British soldiers against typhoid and cholera, who were fighting in different regions of Europe and Asia favourable to the development of cholera epidemics. In a paper stretching over 82 pages, the authors developed the theoretical and mathematical background for calculating the efficacy of vaccines.
This article seeks to explain to the lay reader what these numbers imply and to bring out the differences between efficacy and effectiveness of a vaccine.
Efficacy and effectiveness
At first sight these two terms appear to be synonyms. However, in the world of vaccines and medicine, these two terms are not the same. Efficacy of a vaccine is how it performs under ideal and controlled conditions in a clinical trial (see below). During clinical trials, the outcome of vaccination is compared between a group of vaccinated people and another group given an inactive form of the vaccine (called a placebo). The effectiveness of a vaccine is how the vaccine performs in the real world – that is after the vaccine is approved by the regulatory agencies and you and I are vaccinated.
The efficacy of a vaccine is measured by the manufacturers under ideal conditions in a clinical trial where criteria are specified for selecting and excluding volunteers. These criteria are usually age groups, gender, ethnicity, geographical location and socio-economic standing. If the criteria are specific, then the effects of the vaccine or drug would not be applicable across the population. For example, if the COVID-19 vaccines are not tested on children below 18 years, then the approved vaccine cannot be used on children.
The effectiveness of a drug or vaccine is a measure of how well the drug or vaccine performs in real life, in a diverse population: Fitness geeks and couch potatoes, housewives and nurses, and farmers and office workers. Effectiveness is of relevance to the medical community and healthcare authorities who are treating the patients. Thus, studies on effectiveness would look at to what extent the vaccine is beneficial to the patient to prevent infection.
One may ask, why not simply look at the effectiveness of the vaccine? This is because if the participants in an initial trial of the vaccine are not carefully controlled, then it is difficult to interpret the outcome of the trial. We have many characteristics, which can potentially interfere with the outcome of a trial testing a vaccine. The person volunteering for the trial could be young or old, pregnant or not, a marathon runner or an average person and smoker or non-smoker. Thus, the volunteers selected for the trials are very similar within their groups with many criteria to exclude persons who could confuse the results (for example, an unhealthy person with other diseases would be excluded).
Efficacy of a vaccine asks the question ‘Does the vaccine work under ideal conditions?’ On the other hand, a study on the effectiveness of the same vaccine asks the question ‘Does vaccination work in the real world?’
Clinical trials
Under normal circumstances, vaccines take many years of research and testing to be approved. The COVID-19 pandemic was unprecedented, and pharmaceutical companies embarked on a race against time to produce safe and effective vaccines. The genome of this coronavirus, which was discovered by Chinese scientists, in January 2020, was a major contribution to the development of the vaccines. At the moment there are 94 vaccines being tested on humans in clinical trials, 32 of which have reached the final stage of Phase 3 testing.
To obtain approval for a vaccine, the vaccine manufacturers go through a prescribed process to ensure that the vaccine is safe. All the countries have a national drug approval agency, who should approve the use of a drug or vaccine in that country. The Food and Drug Administration (FDA) in the United States is an important regulatory agency, which has stringent criteria to approve medicines and drugs. In Sri Lanka, it is the National Medicines Regulatory Authority. COVID-19 vaccines are also assessed and approved by the WHO.
Initially, the vaccine is tested on cells in the laboratory and then given to animals, usually mice or monkeys. After this, if the mice or monkeys are happy, human volunteers are recruited to conduct the clinical trials, which is done in three phases. In the first phase, the vaccine is tested on a small group of people to determine the safety, dosage and ability to stimulate our immune system. If this is confirmed, the vaccine then moves into the Phase 2 stage where the safety of the vaccine is tested on hundreds of people who are split into different groups. Once these trials are successful, the vaccine moves to the final Phase 3 trials. Here thousands of people are recruited as volunteers. For the Pfizer-BioNTech vaccine there were over 40,000 volunteers, above the age of 16, from different countries. This trial is more comprehensive, with the volunteers belonging to different age groups, physical fitness, ethnicities and geographical locations. The volunteers are divided into two groups. One group gets the real vaccine while the other group gets a fake vaccine or placebo (the syringe has just water). The volunteers would not know if he/she is getting the vaccine or a placebo and neither do the nurses and doctors giving the vaccine. This is called a double-blind clinical trial. Thus, no one knows, except those conducting the trial, who was vaccinated with what.
After some time, the volunteers, who fell sick with the coronavirus, are PCR tested to confirm if they are COVID-19 positive. The scientists will be on the lookout for any side effects of the vaccine; if they find any cause for concern the trial can be stopped temporarily to conduct investigations and remedy the problem. If the scientists are not satisfied, the trial would be abandoned. Once the results are in, the calculations are done, and all the details are submitted to the regulating authorities. The regulators would ask the manufacturers more questions and once they are satisfied, approval is given to manufacture and market the vaccine. To accelerate the process, such as now during the COVID-19 crises, Phase 1 and 2 may be combined and run in parallel.
Calculating efficacy
The calculations involved are quite simple once the data is collected. Let us assume that 50,000 volunteers were recruited for the vaccination trial. Half were given the vaccine and the other half a placebo. Let us assume that of the 25,000 who received the vaccine, 10 persons were infected, and of the other 25,000 who received the placebo, 200 were infected. Although the numbers of people infected are small, those in the placebo group are 20 times larger (see Table). The researchers are concerned with the relative risk between the groups. This is called the efficacy of the vaccine.
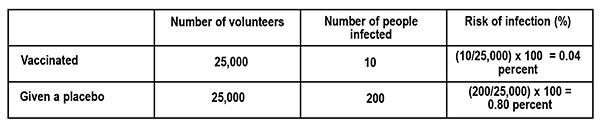
The risk of infection is calculated as follows.
What is the difference in the risk of infection between the vaccinated group and those who got the placebo? From the table this is, 0.80 percent – 0.04 percent = 0.76 percent.
Thus, the vaccine reduced the risk of infection by 0.76 percent, which looks quite small. This is what would happen if we are vaccinated. To understand this in terms of the risk of infection, if none were vaccinated, we look at the ratio of the Reduction in Infection (0.76 percent) to the Risk of infection (0.80 percent – those who got the fake vaccine). This is the Vaccine Efficacy (VE).
VE percent = Reduction in infection ÷ Risk of infection = 0.76 ÷ 0.80 = 95 percent
If this is still confusing, let us see what it means in a population of 100,000 persons who are vaccinated with a vaccine of 95 percent efficacy, and exposed to the virus. From the table above, the risk of infection for the vaccinated population is 0.04 percent, which translates to 40 persons (0.04 percent x 100,000). That is, we can expect that 40 persons would fall ill with an infection by the coronavirus and the rest of the vaccinated people may not develop an infection at all or develop an asymptomatic infection (you are infected but do not show symptoms) or get a mild disease.
(This example of calculating Vaccine Efficacy is adapted from an article by Dashiell Young-Saver in the New York Times of December 13, 2020, where the above calculation is explained in detail for students.)
What does efficacy mean?
The efficacy of a vaccine refers to two aspects. The first is how many of us are protected by the vaccine if we are exposed to the virus; this is given by the percentage. The vaccine also refers to different disease conditions it is capable of preventing. This could be causing an infection, mild disease, severe disease, hospitalisation, or death. This information can be found if one looks carefully at the statements issued by the vaccine manufacturer and regulatory agencies. For example, the statement by Pfizer-BioNTech states: Pfizer-BioNTech COVID-19 vaccine, BNT162b2, was 91.3 percent effective against COVID-19 (symptomatic cases of COVID-19), measured seven days through up to six months after the second dose. The vaccine was 100 percent effective against severe disease as defined by the US centres for Disease Control and Prevention (CDC), and 95.3 percent effective against severe disease as defined by the US FDA.
The efficacy of a vaccine (VE) is the relative reduction of being infected, if we are vaccinated, compared to the placebo or unvaccinated group. If the vaccine is perfect, then the risk of being infected is totally eliminated, so that VE = 1 or it is 100 percent. On the other hand, if there is no difference in the number of people infected between the two groups, the vaccine has no efficacy, or it is zero. Even with a perfect vaccine, our capacity to acquire an infection is determined by our age, health and immunity status.
In short, efficacy is a statistical measurement based on clinical trials of the vaccine’s ability to prevent infection. The volunteers taking part in the trials are not a perfect sample or representative of the real world (for example, children and sick people do not take part). Is there a lower limit for the efficacy of a vaccine to be accepted? Under the present circumstances, the FDA said it would consider granting emergency approval if the vaccines showed even 50 percent efficacy; the vaccines that have received approval now show an efficacy of over 90 percent.
Effectiveness
The effectiveness of the vaccine tells us how well the vaccine is performing among the population, in the real world, to prevent infection. The effectiveness of the vaccine depends on the impact it makes on society. After vaccination our immune system is primed to combat the coronavirus, reducing the multiplication of the virus in our body. This will gradually slow down the spread of the virus as more and more people are vaccinated. In other words, it is important that most if not all the people are vaccinated to have a large impact on the spread of the virus in society. Good examples are the smallpox vaccine, which completely eliminated the smallpox virus, and the polio vaccine, which has almost wiped out the polio virus except for a few small pockets in Pakistan, Afghanistan and Africa. Thus, the effectiveness of a vaccine looks at the medical and societal importance of the outcome.
Here is the above in a nutshell. The percentage numbers given with a vaccine refers to its efficacy – its ability to prevent an infection developing into a serious condition, determined under controlled clinical trials. Vaccines do not prevent infection – they prevent the infection from developing into a severe disease. Once we are vaccinated, our immune system is activated. If we are infected by the coronavirus, the virus has a small window of time to multiply, before it is eliminated by our immune system. This means we can release virus particles from our body, but much less than if we were not vaccinated. The message is we should get vaccinated with the first available vaccine and still wear our masks when going outside, even if we are vaccinated. The chances of ending up in a hospital is low and the chances of ending up in the ICU is very low. There is always a chance.
‘Tis impossible to be sure of anything but Death and Taxes (Christopher Bullock, 1716).
(M.C.M. Iqbal is Associate Research professor, Plant and Environmental Science, National Institute of Fundamental Studies, Hanthane Road, Kandy, and can be reached at iqbal.mo@nifs.ac.lk)
References
Zimmer, C. New York Times Nov. 20, 2020. Two companies say their vaccines are 95 percent effective. What does that mean?
Haelle,T. Association of Health Care Journalists. October 22, 2020. Know the nuances of vaccine efficacy when covering Covid-19 trials. https://healthjournalism.org/blog/2020/10/know-the-nuances-of-vaccine-efficacy-when-covering-covid-19-vaccine-trials/
Greenwood, M., & Yule, G. U. (1915). The Statistics of Anti-typhoid and Anti-cholera Inoculations, and the Interpretation of such Statistics in general. Proceedings of the Royal Society of Medicine, 8 (Sect Epidemiol State Med), 113–194.
Food and Drug Administration, US Department of Health and Human Services. https://www.fda.gov/media/139638/download
Opinion
YUGA PURUSHA Rabindranath Tagore

Where the mind is without fear
And the head is held high
Where knowledge is free
Where the world has not been broken up
Into fragments by narrow domestic walls
Where words come out from the depths of truth …
Into that heaven of freedom, my Father,
Let my country awake
That was not a man ‘for all seasons’ (who are plentiful) but a man for the ages, writing those words in this kali yugaya.
Do you hear them? Now? Now, as ever, as everywhere?
Fifty years ago, I wrote commentaries on each poem in Gitanjali, from which those lines are taken. They were a kind of ‘crib’, paid for by an early tutory, Atlas Hall, which sort of prepared students for examinations at tertiary level here and in London. One might note that Gitanjali and other works by writers in South Asia (other than those touted by spurious academics as ‘post-modernist’ and ‘post-colonial’, – read ‘pro-colonial’) – have long been sent out of the window of classrooms in this country.
The immediate occasion that called for these comments was the presentation of a selection of songs, from Tagore’s extensive body of work, at the Wendt last Monday. It was by the foremost exponent today of robindra sangeeth, Rezwana Chowdhury Bannya of Bangladesh & Santiniketan (yes, that sounds as if Santiniketan is a nation by itself). In a singularly happy namaskar towards each other, it was co-hosted by the High Commissions of Bangladesh & India. The fact that both have adopted Tagore’s songs as their national anthems may be indicative of ‘the breaking down of narrow domestic walls’. ‘The Partition of Bengal’, first attempted by the British over a hundred years ago, failed because the people, Tagore active among them, did not want it. Four decades later they, the Brits again, succeeded in rebuilding that wall though it remains porous. As Sarath Amunugama observed, in a felicitous address in which he referred both to ‘the partition’, and to national anthems, and as is well known here, Ananda Samarakone’s namo, namo matha was inspired by his stay at Santiniketan. In the 1930s to the 1960s the latter connection has vitalised our dancing, singing, ‘music-making’ and our knowledge of theatre.
A somewhat hilarious outcome of the latter occurred about ten years ago at the Tower Hall, when Suchitra Mitra, whose name would for the foreseeable future be inextricably associated with robindra-sangeeth, invited our ‘old boys’ of Santiniketan to come up and join her in their school song. Most of them had lost the words and more than there seemed to be of them had lost their voice, leaving Suchitra Mitra up there encouraging and reprimanding them like a Montessori teacher.
And now we have, before our astonished gaze, a Cricket World Cup with loads of some kinds of drama, including a battle royal among three South-Asian giants of that English game with the sort of statutory-leaders of India, Pakistan and Sri Lanka present, polishing or twirling moustaches and waving gaily in the general direction of our millions of hoi polloi via TV cameras.
Sorry, yuga purusha, no trace of awareness around. So how could you and all of us whom you left behind (not that it could any longer matter to us as it did not to you), expect guilt?
The special issue of INDIA Perspectives (IP) that marked this occasion is a handsome work. The IP journal has always been a high-quality production but this was a revelation. Specialists in each area of Tagore’s interests and activities have contributed articles on his views on schooling, theatre, painting, religion, nationalism and internationalism, science, rural economics and so on, each from his/her perspective. What follows is drawn from that work.
Although he and Gandhi were friends and, says Amartya Sen, he had popularised the appellation Mahatma for Gandhi, Tagore had seen that the chakra was not the route to India’s future. There could be many views on that: Tagore may have overlooked its symbolic value or significance. After all, the bottom-line is that the European tribes became rich by pillaging the rest of the world and rendering those people poor. The textile industry in England, for instance, ‘developed’ by destroying the textile industry in Bengal; the methods adopted were various, the most direct being that of chopping off the fingers of the weavers. Tagore should have been aware of that.
The brutality of the British ‘raj’ was not unknown to him. Following the massacre of over 1,000 unarmed people at a gathering at Jaliawallah Bargh by a Brigadier (named Dyer) Tagore returned a ‘knighthood’ ‘bestowed’ on him by their monarch. A dozen years later, the oh-so-valiant Brits followed up the massacre at Jaliawallah Bargh by, in Tagore’s words, ‘a concerted homicidal attack, under cover of darkness, on defenseless prisoners undergoing the system of barbaric incarceration’. Any other examples, anyone?
Tagore had been an inveterate traveler and the questions that arise in ‘looking inwards and outwards’ tend to remain unresolved. He had foreseen that ‘science’ would be prostituted, that it would not serve the world community of living things, that it would become a man-made calamity: ‘Science is at the beginning of the invasion of the material world and there goes on a furious scramble for plunder. Often things look hideously materialistic, and shamelessly belie man’s own nature.’
Nevertheless he seems to have retained golden visions for what it was going to do: ‘But the day will come when some of the great processes of nature will be at the beck and call of every individual and at least the prime necessities of life will be supplied with very little care and cost’. (We have seen how Monsanto, Del Monte and fellow predators, have set about doing that). ‘To live will be as easy to man as to breathe, and his spirit will be free to create his own world.’ He was fortunate indeed in not being around to witness how the country he was born in and which had nourished his creativity has gone in the pursuit of command of the great processes of nature (and of her neighbours). Besides, the mega-mega weddings, etc., we are witness to the operations of an imperium hell-bent on evicting people from the lands, waterways and beaches that ‘the market’ covets.
How such a culture of science would choose to help the sick or, just a step further for such minds, to make the healthy ill, or, indeed, how such ‘science’ would be used to create, in Ralph Pieris’s term, ‘illth’ (not ‘wealth’), did not quite come to pass in his lifetime. Since his passing, we share a common experience of ‘patents’ on traditional medicines, including the most ubiquitous and widely / wisely used, kohomba or neem, of kotala himbutu and many others, acquired via ‘laws’ constructed by the ‘developed’ people aforementioned, and India’s experience in developing an antidote to the AIDS virus. They affirm the validity of Tagore’s ‘gut reaction’ to where ‘science’ may take the world and has indeed taken it.
Forty years ago Senaka Bibile initiated the construction and adoption of a formulary that reduced the number of drugs required in this country by some 80% and identified them by their generic name, and battle was joined. (Senaka was eventually eliminated/killed by a mercenary, from this part of this world, of Big-Pharma). That entity, Big-Pharma, has acquired control not only over the production of drugs and their marketing but over the entire range of activity that relates to health-care – systems of ‘referral’ and lab tests where such weren’t needed, so with hospitalisation or indoor treatment usually with yet more ‘tests’, ‘prescription drugs’, ‘insurance’ from an ‘approved’ company of blood suckers. Its control is most scandalously evident in the USA and includes a species of corruption that Tagore could not have conceived of. (robindra–sangeeth does not address such yet-to-be reality, nor do his plays and paintings). When Big-Pharma got their obedient servants in the USA administration to send in marines to force Bangladesh to allow their drugs in, the government and the people of Bangladesh, all honour to them, physically ‘repelled the boarders’.
Tagore lived in and came to terms with a changing world, and he responded to all of what he saw in terms that had not occurred to his contemporaries anywhere in ‘the known world’. There were others of course who had a like foresight. Though too numerous to mention here, I should think that Blake and Whitman belonged among them, – as did such great poets as Bharathari from centuries ago, and Subramaniam Bharathi, consigned to a pauper’s grave, from yesteryear. So many more through all the hundreds and thousands of years that don’t quite make up a kalpa.
We learn through the IP that Tagore’s name had been put up for the Nobel prize by a single member of the Royal Society, T S Moore, while 97 other members had collectively recommended Thomas Hardy. The Swedish Academy had picked Tagore out of 28 nominees. In a telegram conveying his acceptance of the award, Tagore expressed his appreciation of ‘the breadth of understanding which has brought the distant near, and has made the stranger a brother’. In these times, Sarkozy, Cameron and their ilk seem intent on making strangers of brothers.
A fallout of the instant fame it brought had been a loss of privacy (as Garcia Marquez and others discovered many decades later) and of the use of his time to get on with his work. Gitanjali was for the most part a rendering into English, by the poet himself, of his songs in Bangla. Translating a novel, short story or a play is no easy matter (as, with respect to Sinhala works, Ashley Halpe, Lakshmi de Silva, Vijitha Fernando et al could confirm). Hemingway had found the great Russians unreadable till he came upon the translations by Constance Garnett. Translating poetry is infinitely more difficult, (as Ranjini Obeyesekere and Lakshmi have shown) and Tagore was hounded by admirers to translate more of his work into English. He was called on to make his poetry accessible to those who had only English. His poems have since been put into English; among them, an effort I liked, a whole volume, was titled ‘I will not let you go’. Simply put, the title poem will not let you go.
Nevertheless, the task of translating works in other south Asian languages, to begin with, into Hindi, Bangla and Urdu and the other way is one that needs attention. Bangla has the second largest numbers of speakers in South Asia after Hindi – about two-thirds the number of Hindi-speakers. Bangladesh might consider setting up a kind of clearing house for such work, perhaps with SAARC support and located perhaps, at Silaideh, around Tagore’s ancestral land in Bangladesh. Maybe, as Tagore’s examples show, ‘start small’ would be a good approach.
On matters that have to do with ‘religion’, Tagore’s activities may be seen as being eclectic. He was a member of Brahmo, (of which Satyajit Ray and his father’s family were members), which took the Upanishads for text and had no truck with caste-orders of ‘Hinduism’ including the rationalization for it given in the Gita. He admired Sufism, presented a ‘Christothsava’ akin to Christmas, wrote on ‘Devotion to Buddhism’. His view on Siddhartha Gautama was: ‘This wisdom came, neither in texts of scripture, nor in symbols of deities, nor in religious practices sanctified by ages, but through the voice of a living man and the love that flowed from a human heart.’ The concept of nirvana had not attracted him and in that sense his perception of Buddhism seems to have been closer to that of the northern form than to the Theravada familiar to us here and in south-east Asia.
As with his experiments in theatre, where he moved away from the westernised urban mode to the folk-inspired dance-drama, so with music and song he moved away from the classical raag to folk music. That is a trajectory that our musicians should explore. He drew from other cultures – among the vibrant renderings given by Rezwana Chowdhury Bannya was one that gave a celebratory edge to ‘Ye banks & braes o’ bonnie Doon’.
My first encounter with robindra sangeeth occurred in Dhaka at the home of Mohamed Sirajuddin. When the late Prof. P P G L Siriwardena introduced us, Siraj exclaimed, ‘We are batch-mates’; what he meant was that he had joined the CSP (Civil Service of Pakistan) around the same time as I joined the CCS. As Secretary for Rural Development he did much to support cottage industries in Bangladesh and was familiar with our experience in that field. He invited artistes he valued, some, to my ears, at master level in robindra sangeeth, to perform at his place. I was struck by the variety of those who turned up to listen; there were friends, people from down – or off – the road, the Governor of the Central Bank, Ministers, colleagues … It reminded me of the glory days at Chitrasena’s in Kollupitiya. In an environment that seemed designed for chamber music, those songs sank into my heart. Among those who sang were a young couple who were TV stars but gave tribute to a middle-aged man, Farook, who was a master. Yes, robindra sangeeth, does need the male voice.
As Rezwana mentioned, delicately, as ‘in passing’, a problem that arises in appreciating such songs is that they are more sadly incomplete for the listener who has no Bangla than the emotions they do convey regardless. The affinity between Bangla and Sinhala is well known. (Some twenty years ago I sent a farmer from Berelihela, off Tissamaharama, to Dhaka for extended chats with fellow farmers from Asia and the Pacific. When I myself got there a few days later on allied business, I found that he had communicated very well indeed with people there in the only language he knew: his own). The present moment seems to offer an excellent opportunity for the High Commissions of Bangladesh and India to harness the active support of our government to set up an infrastructure for making Bangla accessible to our people. If, in these sort-of ‘market’ days a further incentive is required at this end, policy makers should be aware that workers and managers from here have contributed much to the resuscitation of a textile industry in Bangla that had been of an unparalleled excellence through the centuries.
by Gamini Seneviratne
Opinion
More about Premadasa

In an article published in The Island of 01 May, Rohan Abeygunawardena has paid a glowing tribute to R. Premadasa. It is true Premadasa, as a man from a humble urban working class, was ambitious, and to boost his personal image he targeted the rural and the common man, marginalised by previous regimes. He set up projects to satisfy these folks and selected his own staff to carry out his orders to achieve what he desired. He got rid of those who were sticking to rules and regulations.
One such case is, J .R. Jayewardene brought in previous prestigious Civil Service officers to revamp the fading public service, and one such was the illustrious Chandi Chanmugam, as Secretary to the Treasury. He was called up by Premadasa and requested to provide funds for a welfare project and when he explained the difficulties, he was bluntly told that he (Premadasa) could find an officer who could make the funds available. In keeping with the traditions of the CCS, Chanmugam tendered his resignation. The vacancy was filled by R. Paskaralingam. When Secretaries questioned about funds, Paskaralingam, who chaired the Development Secretaries Committee, would say, “This is bosses orders, find the funds somehow. ” How the Secretaries provided funds is another story.
The next three projects to boost his image at government expense were the mobile office programme, the housing programme and Gamudawa.
As Assistant Secretary to the Ministry for Power and Energy, I was assigned to conduct the mobile service. As far as I could remember, the first Mobile Office was held in the Yapahuwa Electorate, in a village called Badalgama. The previous day, I rang up the area engineer and asked him to meet me at the school building, allocated for the Mobile Office, and to inform the UNP party supporter, who was to find accommodation for my overnight stay. When I arrived, the Area Engineer was there with men to make arrangements for the mobile office. Then two officers from the Presidential Mobile Office Division walked in and inquired as to why I had not hung a picture of Premadasa as he wanted his picture prominently displayed at Mobile Offices. When I said that I had no picture, they rushed back and came with a beautifully framed picture and hung it on the wall.
The following day, before going to the Mobile Office to take an oath, I went to my office to find that someone had garlanded the picture. It was later found that the clerk, who accompanied the area engineer, had overheard the conversation, knowing Premadasa’s whims and fancies.
The work started and as usual. Premadasa visited all offices and when he came to mine, I greeted him in the oriental fashion but his eyes were directed towards his picture and a beam of smile crossed his face. When leaving he said, “Carry on the good work.” Since then at every Mobile Office, I arranged for a special event for him to attend, such as the opening of a rural electrification project.
Gamudawa: This project was similar to the presidential mobile service. There was a variety show organised by the UNP supporters, and crowds dispersed happily. When the Gamudawa project was to be started, a request was made by the Presidential Secretariat to supply generators as the sites selected were far away from the transmission line. The then Chairman of the CEB, Prof. K. K. Y. W. Perera, who was also the Secretary to the Ministry for Power and Energy, politely replied requesting a payment to meet at least the cost. There was no reply and when I visited the Gamudawa held in Wellawaya, I saw CEB men operating the generators. On my return, I reported the matter to the Secretary to the Ministry and also the General Manager, CEB. They said that they were aware but remained silent.
At the first staff meeting, after the 1988 presidential election, Premadasa said, “Carry out my orders and those who do not agree could find other places.”
This was the start of deterioration in the power and energy sector. He brought in his own staff and the once well-managed sector fell into disarray. Premadasa removed Prof. Perera from the post of Chairman, CEB, and the Workshop Engineer, who supplied the generators without the knowledge of the management, was appointed Chairman, CEB, a reward for carrying out illegal orders! Having been in the state service for 40 years, I walked out happily without a farewell party. I took with me only a wooden block, on which my name was printed, and the Lion Flag, which I displayed at Mobile Offices.
President Premadasa also ordered that all policemen in the Eastern Province, surrender to the LTTE, with their weapons. The LTTE killed all of them, numbering over 600.
G. A. D. Sirimal
Boralesgamuwa
Opinion
Postmortem reports and the pursuit of justice

A serious debate has erupted following a postmortem examination conducted on the body of Ranga Rajapakshe, who was found dead in his garden.
The controversy has arisen as Rajapakshe, an Assistant Director in the Finance Ministry, had been suspended over the diversion of 2.5 million dollars to a fraudulent account. Although the cause of death (COD) is obviously cardiorespiratory failure due to severe haemorrhage (loss of blood), whether the two cut wounds on his legs and on his left wrist were self-inflicted or caused by an external agency is what has led to this raging controversy.
A four-member ‘regional’ expert forensic panel (EFP) was appointed supposedly by the Secretary, Ministry of Health. The Judicial post mortem report was submitted within 24 hours. Many questions have risen as a result. Whether the expert forensic panel looked into all aspects of the death – and not only the injuries in the body of the deceased — has become a moot point.
Was the death due to self-inflicted cut injuries, i. e. suicide? Or, were they inflicted by another or others? If so, it becomes homicide or murder. If there have been any deficiencies in the procedure adopted by the expert forensic panel, whether they are errors, negligence or deliberate is what is reverberating on the social media and the public spaces.
One important point has to be mentioned at the outset. The JPM Report is still not in the public domain. Whether it would remain a privileged communication limited to the judiciary remains to be seen. Hence, none can come to definitive conclusions on the JPM findings – except judicious, informed speculation.
Judicial Post Mortem Examinations: Are they prone to error, negligence or deliberate falsification?
History tells us that all three of the above are possible. The fourth possibility is that it is none of the three above, but a legitimate, academically defensible difference of opinion. Neither medicine, nor forensics is an exact science.
Error
A cursory glance at information on the Internet gives us a reasonable overview of the issue of error. Of them, I quote only those that may be relevant to the issue at hand.
(1) Errors in post-mortem examinations can arise from procedural oversights, misinterpretation of findings, or lack of expertise, with major diagnostic error rates ranging from 8% to 24%.
(2) Common mistakes include misinterpreting postmortem changes as injuries, missing findings due to incomplete examination, and failing to secure the chain of custody.
(3) Incomplete Examination: Failing to examine all necessary body cavities or failing to perform histology/toxicology.
(4) Misclassification of Death Manner: Incorrectly labelling a death as natural vs. unnatural (e.g., suicide vs. homicide) due to overlooking evidence or biased interpretation.
Causes of Errors
(1) Systemic Issues: Heavy workloads, lack of specialised training, inadequate equipment, or poor communication between investigators and pathologists.
(2) External Pressure: Influences from law enforcement, media, or families that can bias the investigation.
(3) Inefficient Techniques: Relying on delegated assistants for vital dissections or conducting superficial examinations.
The above would suffice to give us an idea about lacunae and deficiency in JPM examinations that could lead to error. Those interested could go into the plethora of academic articles on this subject of error in JPMs.
Did any of the above lead to an outcome of error in the conclusions of the JMP Report by the expert panel?
Negligence
Negligence involves critical and serious errors that are inexcusable. These include inadequate body examination, failed scene investigations, missed evidence and speculative, premature reporting. These shortcomings can hinder legal proceedings, obscure causes of death, and lead to wrongful conclusions, with studies identifying major procedural errors, including failure to identify injuries or misinterpreting pathological findings.
We have no information whether the EFP had done a detailed site visit.
Deliberate falsification
Deliberate falsification or fraudulent autopsy reporting involves the intentional alteration of findings, documentation, or conclusions to misrepresent the cause or manner of death.
This misconduct can take many forms, including covering up homicide, misrepresenting police actions, or protecting influential individuals.
Forms of Deliberate Falsification include modification of Conclusions due to Forensic pathologists facing coercion from police, politicians, or families to change a homicide to an accidental death or natural causes. Intentional Neglect of Evidence: Failing to document injuries like strangulation marks or bruises to support a fabricated narrative of natural death. Issuing misleading or untrue post-mortem reports constitutes “serious” professional misconduct that is punishable by law.
There is absolutely no evidence that deliberate falsification has occurred in this case. But what I have attempted to inform the readers of is that such situations are well known.
The celebrated Sathasivam case illustrates the earliest instance in Sri Lanka, in which there was conflicting forensic evidence from two highly eminent forensic professors. Professor GSW de Saram, the first professor of forensic medicine, faculty of medicine, of the then University of Ceylon and JMO, Colombo was the most pre-eminent forensic expert in Ceylon who gave evidence for the prosecution and Sir (Prof.) Sydney Smith, world renowned professor of forensic medicine, University of Edinburgh who gave contrary forensic evidence on behalf of the defence. This conflict in the forensic evidence was a key factor that resulted in Sathasivam’s acquittal
I list below, a few JPM discrepancies and conflicting JPM reports that are now in the public domain in the recent past in Sri Lanka:
1. The death of a student at the University of Ruhuna raped and killed on the Matara beach, considered a suicide when circumstantial evidence indicated thugs of a well-known politician were involved in the incident. I was on the academic staff of the faculty of Medicine, University of Ruhuna at that time and came to know several details that had not come into the public domain.
2. The conflicting PM reports on the “disappearance” of the kidneys of a child at LRH, which was originally given as a medical death and later judgement given as a homicide. The child’s good kidney had been removed when the nephrectomy had to be done on the damaged kidney.
3. The infamous JPM report first given on Wasim Thajudeen’s killing. This falsification was done by a very senior JMO.
4. Lasantha Wickrematunga’s death, which was originally attributed to shooting but subsequently found to be due to stabbing with a sharp implement.
5. The RTA death of a policeman on a motorcycle (his wife and children were also seriously injured) in Boralesgamuwa due to the drunk driving by a female specialist doctor. The first JMO report stated that the doctor had not been under the influence of alcohol until CCTV evidence was presented to the Court that showed her drinking in a club that night. The police informed Court that the breathalyser test had confirmed that the doctor was under the influence of alcohol.
These are some of the well-known instances that there had been conflicting JMO reports. Furthermore, there have been several JMO reports where death in police custody was falsely documented in the JPM or JMO reports to safeguard the police involved in torture.
I know of one case personally, where a doctor from Nagoda Hospital, Kalutara was hauled up by the Sri Lanka Medical Council (of which I was a member for 10 years) for falsifying his JPM report of a death of a young man in police custody to safeguard the policemen concerned.
Why do JMOs falsify JMO reports?
Based on reports and studies, primarily focusing on the context of Sri Lanka, allegations of false or misleading judicial medical reports by Judicial Medical Officers (JMOs) arise from a combination of systemic, ethical, and external pressures rather than a single cause.
Reports indicate that instances of faulty reporting often stem from several factors. The main factor being political and external influence. These are likely in high-profile cases; JMOs may face pressure to tailor reports to suit the interests of powerful individuals or to minimize the culpability of suspects.
It has been seen that some reports are deemed erroneous or contradictory due to negligence, improper reporting procedures, or a lack of understanding of the ethical responsibilities of their role as JMOs. The police sometimes exert influence to speed up investigations, leading to “shortcuts”, where evidence is not properly scrutinised, or reports are tailored to support a premeditated narrative rather than scientific findings.
To be fair by JMOs, it must be said that false history or narratives given by victims and or perpetrators mislead the JMO. Victims or suspects may provide false history during the medical examination to protect themselves or to misdirect investigations.
The dearth of experienced forensic specialists can lead to inexperienced officers handling complex forensic cases. It has been the practice in many instances that Magistrates make specific requests that the PM examination be transferred to an experienced and senior forensic expert.
The subversion of justice is not limited to our part of the world. It happens everywhere. The judiciary, the legal and medical professions can work together to deliver justice to the impoverished and unempowered masses.
by Prof. Susirith Mendis
susmend2610@gmail.com
-

 News4 days ago
News4 days agoMIT expert warns of catastrophic consequences of USD 2.5 mn Treasury heist
-

 News6 days ago
News6 days agoCJ urged to inquire into AKD’s remarks on May 25 court verdict
-

 News7 days ago
News7 days agoUSD 3.7 bn H’tota refinery: China won’t launch project without bigger local market share
-

 Editorial4 days ago
Editorial4 days agoClean Sri Lanka and dirty politics
-

 Editorial7 days ago
Editorial7 days agoDeliver or perish
-
 Opinion6 days ago
Opinion6 days agoSecurity, perception, and trust: Sri Lanka’s delicate balancing act
-

 News2 days ago
News2 days agoSLPP expresses concern over death of former SriLankan CEO
-

 Editorial3 days ago
Editorial3 days agoThe Vijay factor













